REPORT UNSAFE LASER LIGHT SHOWS
Responsible laser show producers will keep beams from entering audience areas, or will ensure that audience-scanning beams are at eye-safe irradiance levels. Unfortunately, it is not always possible to tell by eye, or from a video, whether audience beams are too powerful.
It is better to be safe than sorry. Therefore, if you have concerns about the safety of any laser display, contact the venue, the laser company (if you can find out who did the show) and also the appropriate government agency(ies).
Contact the laser operator, laser company, venue managment, local police, fire department, state radiological/laser regulator, FDA (see below) or other persons with authority and control over the lasers if you feel that there is an immediate danger.
Note that many of these persons or institutions may not have laser safety knowledge, so you may have to explain the hazards sufficiently for them to take action. You may find that no action is taken, or action is taken too late to be effective. You still can report the unsafe and/or illegal show afterwards to the appropriate authority(ies).
For large-scale tours, an internet search for terms such as “Joe Artist Band 2017 tour lasers production” might turn up a news article, press release or other source with the name of the laser company.
For example, ILDA received a complaint in September 2017 regarding a major artist’s concert. A search turned up an article about the tour in industry magazine PLSN, which listed the major lighting, sound and production providers — including the name of the laser company. (It turned out not to be an ILDA Member.)
A further internet search then can turn up contact information for the company. Let them know about any problem or concern.
The following is from a November 2021 FDA presentation to ILDA, regarding how to report unsafe shows:

Here is a clickable link: https:www.fda.gov/medical-devices/reporting-allegations-regulatory-misconduct/allegations-regulatory-misconduct-form
FDA has an emergency telephone number, staffed around the clock, for “an FDA-regulated product that requires immediate reporting.”
For non-emergencies, to voluntarily report a suspected problem or injury, you can directly report to the FDA by completing and mailing the Accidental Radiation Occurrence Report form.
ILDA invites reports of unsafe or illegal shows. This helps to get an idea of the number of such shows.
Plus, if the show was done by an ILDA Member (see here for list), you can also file an Ethics Complaint. ILDA can investigate and can take action within the association.
However, any serious problems or concerns should first be addressed with the laser operator, laser company, venue management, and government regulators. They will have the authority and power to stop unsafe or illegal laser shows.
ILDA as an organization has no authority or power to stop unsafe or illegal shows. For this reason, anyone who wants a show to be inspected or shut down must contact the appropriate authority(ies). ILDA does not do the contacting for you.
It is better to be safe than sorry. Therefore, if you have concerns about the safety of any laser display, contact the venue, the laser company (if you can find out who did the show) and also the appropriate government agency(ies).
- For example, in the U.S. contact the FDA's Center for Devices and Radiological Health, and your state laser agency.
- In the U.K., contact Public Health England and the local authority for approving shows.
- Links for agencies can be found on this website’s Links and Resources page.
If you feel there is an immediate danger
Contact the laser operator, laser company, venue managment, local police, fire department, state radiological/laser regulator, FDA (see below) or other persons with authority and control over the lasers if you feel that there is an immediate danger.
Note that many of these persons or institutions may not have laser safety knowledge, so you may have to explain the hazards sufficiently for them to take action. You may find that no action is taken, or action is taken too late to be effective. You still can report the unsafe and/or illegal show afterwards to the appropriate authority(ies).
Finding and contacting the laser company
For large-scale tours, an internet search for terms such as “Joe Artist Band 2017 tour lasers production” might turn up a news article, press release or other source with the name of the laser company.
For example, ILDA received a complaint in September 2017 regarding a major artist’s concert. A search turned up an article about the tour in industry magazine PLSN, which listed the major lighting, sound and production providers — including the name of the laser company. (It turned out not to be an ILDA Member.)
A further internet search then can turn up contact information for the company. Let them know about any problem or concern.
Contacting the U.S. government
The following is from a November 2021 FDA presentation to ILDA, regarding how to report unsafe shows:

Here is a clickable link: https:www.fda.gov/medical-devices/reporting-allegations-regulatory-misconduct/allegations-regulatory-misconduct-form
FDA has an emergency telephone number, staffed around the clock, for “an FDA-regulated product that requires immediate reporting.”
For non-emergencies, to voluntarily report a suspected problem or injury, you can directly report to the FDA by completing and mailing the Accidental Radiation Occurrence Report form.
Contacting ILDA
ILDA invites reports of unsafe or illegal shows. This helps to get an idea of the number of such shows.
Plus, if the show was done by an ILDA Member (see here for list), you can also file an Ethics Complaint. ILDA can investigate and can take action within the association.
However, any serious problems or concerns should first be addressed with the laser operator, laser company, venue management, and government regulators. They will have the authority and power to stop unsafe or illegal laser shows.
ILDA as an organization has no authority or power to stop unsafe or illegal shows. For this reason, anyone who wants a show to be inspected or shut down must contact the appropriate authority(ies). ILDA does not do the contacting for you.
Information to include
- Your contact info — name, address, email, phone, best way to contact you
- Date of your report
- Laser show date, start time and location (city, state, venue).
- Approximate time (or what song, event, etc. was happening) during the show when the problem(s) occurred.
- Name of the artist or event. Also, if you happen to have the name of the producer, the laser company, or other potentially responsible parties.
- Nature of the problem, issue or injury. If an injury, discuss any treatment or medical visits as a result of the laser exposure.
- A timeline, if applicable. For example, “The show started at 7 pm. The lasers came on about 8 pm. I was hit in the left eye about 8:15 pm. At 9 pm I noticed fuzzy vision in the eye.”
- About how far you were from the laser source
- Your seat location or section. If you can include a diagram of the venue seating and stage location — such as from a ticket selling website — this may be useful. For example:
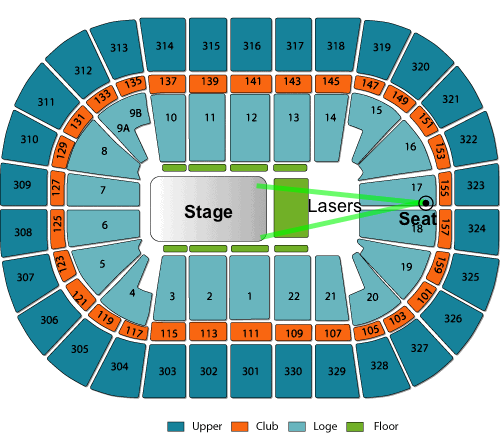
- Whether the laser beams seemed thin/sharp or fat/fuzzy.
- Whether beams were deliberately aimed into the audience — it was part of the show —or whether this was potentially an accidental occurrence
- Color of the beams, both in general (all beams) and the one(s) that caused your problem, issue or injury
- Whether other people had problems such as vision problems, or were commenting on the lasers being too bright, or otherwise seemed adversely affected
- How often beams went into the audience; for example, “off and on over the last hour”, or “only the one time they hit me in the eye”
- Whether there are any photos or videos you (or others) took to show the laser usage. You may want to check YouTube, Instagram, Facebook and similar sources that may have uploaded photos or videos. Provide links for FDA reference, and also download and store the photos/videos in case they are removed before FDA can view them.
- Whether there was any damage to a camera, camcorder or other sensor. (Note: sensors can be damaged at lower thresholds than eyes. Laser damage to a sensor does not automatically mean the laser could also cause eye injury.)
- If the laser exposure seemed to be an accident, was there any action taken such as some or all lasers being turned off?
- Name of any parties that you contacted, either during the event (an usher or security person) or afterwards (emailing a producer or laser show company)
- Copies of any correspondence with other parties
menu